The coronavirus vaccine: what you need to know
We explain what we know about the potential vaccines and what they could mean for people with blood cancer.
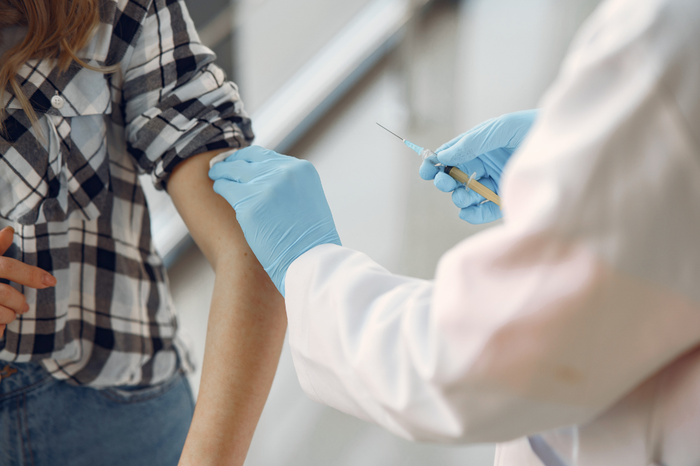
Results from clinical trials of several coronavirus vaccines have been heavily reported in the media over the last few weeks. Although the results haven’t yet been formally published in journals, many of the vaccines have reported high levels of effectiveness in large numbers of people.
For over 200,000 people with blood cancer who have been shielding, a vaccine offers hope of life returning to somewhat normal. In this blog, we explain what we know about the potential vaccines and what they mean for people with blood cancer.
Visit our coronavirus hub
Visit our covid hub to read detailed information about the coronavirus vaccine and blood cancer.
What’s been reported so far?
The Pfizer vaccine
The pharmaceutical company, Pfizer, were the first to release results of its clinical trial, which showed its vaccine to be 95% effective in a study of 43,000 people.
Importantly, this trial looked at people aged 65 and over, whose immune systems are weaker than those of younger people. This is because our immune systems ‘age’ as we do. In this age group, 94% effectiveness was observed.
How exactly does this vaccine work?
The vaccine takes part of the genetic code of the coronavirus and puts it in a vaccine. It’s important to note that this is not a live vaccine.
Once injected, this code tells our cells to produce a protein found on the surface of the coronavirus. Our immune systems respond to this and ‘remember’ what coronavirus looks like.
When encountered again, our body will recognise it, allowing us to respond quickly to avoid becoming ill. The UK has ordered 10 million doses of this vaccine by the end of the year, and an additional 30 million after that.
The Moderna vaccine
After the results from Pfizer, Moderna shared their findings from their clinical trial on a vaccine that uses similar technology to the Pfizer vaccine. This saw 95% effectiveness levels in a study of 30,000 participants. Again, this vaccine is not a live vaccine.
This study involved 7,000 people over the age of 65, as well as people with diseases that put them at high risk of complications from the coronavirus, such as diabetes and cardiac disease. But, as far as we’re aware, this did not include people with blood cancer. The UK has ordered 5 million doses of this vaccine.
The AstraZeneca / Oxford vaccine
This vaccine is made from a modified version of a virus that causes the common cold in monkeys. This vaccine isn’t typically ‘live’ as genes have been removed from the virus, so it is no longer able to cause disease.
Researchers have then inserted an additional gene, which when injected, prompts our bodies to make a protein found on the coronavirus. Although, not a protein that will cause infection. Our immune system should then respond to this, preventing infection in the future.
This vaccine has been shown to be between 60 and 90% effective depending on how doses are administered in a clinical trial of 20,000 people. The UK has ordered 100 million doses of this vaccine.
These are just three of more than 300 vaccines in development, which all take slightly different approaches. The technology behind the Oxford vaccine is different to the Moderna and Pfizer vaccines, and some vaccines may be more suitable for certain groups of people than others.
All vaccines undergo a thorough review by the Medicines and Health Regulatory Agency (MHRA) before they can be given to people. We’re waiting to see what this means for people with blood cancer.

Stay updated with the latest news
Sign up to our mailing list to receive the latest news, research, and ways you can help us beat blood cancer.
Will the vaccine be effective for people with blood cancer?
This question is difficult to answer with a simple ‘yes’ or ‘no,’ as it depends on what vaccine people receive and what type of blood cancer people have. Having said that, we’ve spoken to various blood cancer experts and consultants who would strongly encourage people to have a vaccine (assuming it’s approved by the regulator), as any protection is better than none.
It’s similar to the flu vaccine – every year, many haematologists recommend their blood cancer patients receive the flu vaccine, as they have some immune response, even if it doesn’t work as well as in someone who doesn’t have blood cancer. Some protection is better than none.
As far as we know, no one with blood cancer has received a coronavirus vaccine in these trials. While we think a vaccine is likely to be safe (more on this below), we don’t yet know what level of protection they will offer to people with blood cancer.
However, we know that research is being planned, which will monitor this. This might show that people with blood cancer need an additional ‘booster’ dose of the vaccine. Alternatively, they may receive other treatments to enhance protection against coronavirus.
The Joint Committee for Vaccination and Immunisation (JCVI) has published provisional guidance which says that people who are clinically extremely vulnerable, which includes those with blood cancer, will be prioritised at the same time as the over 70's. After residents of care homes and workers and those ages over 75.
In addition to his, as more people receive a vaccine, the risk of encountering the virus should be lower for people with blood cancer. This is based on initial research, which has shown that the vaccine reduces the chances of someone being able to pass on the virus.
Is it safe for people with blood cancer to have the vaccine?
As mentioned above, and as far as we’re aware, no one with blood cancer has received a coronavirus vaccine in these trials. However, from speaking to clinicians and experts, we think that a vaccine should be safe for people with blood cancer.
This is because the clinical trials that have taken place have involved tens of thousands of people, and none have shown any serious side effects that cause concern. We can also infer things based on how the vaccines are made, and how people with blood cancer have responded to similar vaccines.
Ultimately, it will be down to the MHRA – who are responsible for licensing vaccines – to decide which vaccines are appropriate for which people based on results from clinical trials. We will share this information with you as soon as we have it.
Many people are concerned about how quickly this vaccine has been made.
However, the flu vaccine is made in a very similar timeframe. Every year, a new flu vaccine comes along that is made from scientists taking an informed decision on what strains of flu they think will be prevalent. This is then tested in clinical trials and rolled out to the general population in a similar timeframe to how the coronavirus vaccine has been developed. Therefore, we shouldn’t be unduly worried about the speed.
In addition to this, governments and pharmaceutical companies have invested large sums of money in this research, which has made these trials far more streamlined and supported than they would usually be. This support has meant that the trials have been able to recruit the same number of people as typically done in other trials, in a shorter time.
It’s true that the vaccine hasn’t been around for a long time, but if there were going to be any problems with the vaccine, it’s likely that it would be detected early on in the trial.
We shouldn’t think that the timeframe makes this vaccine any more risky than a typical vaccine.
Ultimately, whether someone chooses to have a vaccine or not comes down to risk versus benefit. More than 200,000 people with blood cancer in the UK have had their lives completely changed over the last year. For them, the benefits of a vaccine could be huge.
Not only could it mean that life could get back to some sort of normality, but it could also potentially be lifesaving. Nothing comes without a small risk, but the potential benefits of getting a vaccine far outweigh the risks of not.
Are there any treatments which might mean that I wouldn’t respond to the vaccine?
Some blood cancer treatments significantly decrease our immune response. These include treatments like rituximab, which can wipe out the B-cells that are responsible for making antibodies. It also includes chemotherapy treatments, like bendamustine.
However, experts we’ve spoken to still suggest that people receiving these treatments should have a vaccine. This is because it may still offer some protection, and some protection is better than none at all.
The only caveat to this is for people who have had stem cell transplants. For these people, the immune system can take time up to a year post-transplant to recover. Particularly for those who have had a transplant from a donor.
In this instance, it’s looking like a coronavirus vaccine would be given a year or so after the transplant, when these people receive their other vaccines. We will keep monitoring this and provide you with updates as and when we get them.
What other research is needed?
We hope that the MHRA will confirm that enough research has been done to be able to offer a safe vaccine to people with blood cancer. We now want to understand the effectiveness in people with different types of blood cancer.
Scientists are planning research that will measure the response people with blood cancer have to a coronavirus vaccine. This should help us to understand whether some people need perhaps an additional dose, or alternative methods to protect them against the virus.
It was recently announced that AstraZeneca are trialling an injection which contains antibodies to fight the coronavirus in people who have impaired immune systems. We’ll keep you posted on this.
Will I have to continue shielding after I’ve had the vaccine?
Unfortunately, we don’t yet know the answer to this question. This will depend on how effective the vaccine is for people with blood cancer, and whether vaccinating the general population can stop people passing on the virus.
This may take some time to understand. We anticipate that the Government would issue guidance to reflect this and ensure people with blood cancer are kept safe.
Will my family be prioritised for the vaccine?
At this stage, we are unsure if people who live with those shielding will be prioritised for vaccination. Current provisional guidance suggests that those who live and work in care homes will be prioritised first, and then people will be prioritised by age group.
However, we have highlighted the importance of this issue to the Government and if your family are invited to have a vaccine, we would recommend that you encourage them to have it.
Where will I have the vaccine?
As it stands, we’re unsure where people with blood cancer would receive a vaccine. We’re seeking further clarification on this and will update you as soon as we know more.
We hope this blog has been helpful in answering some of your questions and we will keep you updated as and when we know more.
With thanks to: Professor Mark Drayson, Dr Graham Collins, Dr Karthik Ramasamy and Dr Matthew Buckland for their insight.
If you've got questions or concerns around the coronavirus vaccine, or about blood cancer in general, please don't hesitate to call or email our free, confidential support team.
