Clinical Trials Support Service
Finding a clinical trial and understanding the complex information to enrol can be overwhelming but we're here to support you and help you navigate through the entire process - whether you're a patient, carer or healthcare professional.
The Clinical Trials Support Service was launched in 2020 as one of the first of its kind in the UK.
We've already helped well over 500 people find out more about accessing clinical trials.
How our Clinical Trials Support Service can help you
Jane, Rachel and Lynne are registered nurses experienced in blood cancers and clinical trials.
They are here to support you and help you navigate through the entire clinical trial process.
How can they help?
- By providing personalised support around understanding if participating in a clinical trial could be right for you
- By helping you understand the clinical trials process, what could be involved, and answer any questions that you have
- Where possible, providing you with a list of clinical trials based on your blood cancer diagnosis, general wellbeing, past medical history and past treatment (if any), that you can then share with your treating team
- Be available to support you throughout your clinical trial journey
- By being there if a clinical trial is not needed now, but may be needed in the future
Please note, that all treatment decisions are made by your treating team, which consists of a consultant and other healthcare professionals. We do not have the authority to make treatment decisions.
Get in touch
Our Clinical Trials Support nurses are available between 10am and 4pm Monday to Friday (except Bank holidays). If you'd like to get in touch, please complete the referral form below.
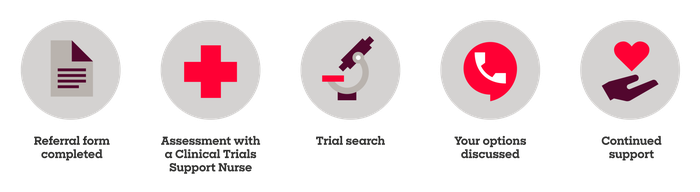
The Clinical Trials Support Service Journey
I was impressed with how personalised the service was, including a conversation with the trials nurse over Zoom, and with the quality of the support that I received.
- Rachel, Clinical Trials Support Service user
Meet the Clinical Trials Support Service Team
Jane, Rachel and Lynne are registered nurses experienced in blood cancers and clinical trials. The team is supported by Leanne, Clinical Trials Data Officer
Meet Jane
Senior Clinical Trials Support Nurse
Jane has over 20 years' experience working in Haematology at the Queen Elizabeth Hospital, Birmingham, with the last 13 years as Senior Haematology Research Nurse within one of Cancer Research UK's Clinical Trials Units. Jane brings this wealth of knowledge and experience to Blood Cancer UK to head up the Clinical Trials Support Services Team.

Meet Rachel
Clinical Trials Support Nurse
Rachel has worked as a Staff Nurse within a variety of cancer care settings, from acute Haemato-Oncology units to specialised stem cell transplant wards. Most recently, Rachel worked as a Senior Staff Nurse/Research Nurse at the Drug Development Unit at the Royal Marsden, facilitating the set up of delivery of first in human clinical trials. She is passionate about improving access to clinical trials for everyone affected by blood cancer.

Meet Lynne
Clinical Trials Support Nurse
Lynne has worked within the specialities of oncology and haematology for over 30 years. The majority of these roles have included hands on clinical work and one to one care with patients. Most recently, Lynne was a Clinical Nurse Specialist providing care and clinics to people receiving treatment for lymphoma and blood disorders. Lynne is excited to be working within a team that can give time, support and information to help people affected by blood cancer make informed decisions about their care.

Meet Leanne
Clinical Trials Data Officer
Leanne is the Data Officer within the Clinical Trials Team. She has over 10 years experience working in data and technical roles in both public and private sector organisations. She is passionate about data and more importantly how data can be used to help people. Leanne discovered her passion for the Charity Sector when previously working as a Fundraising Data Officer, where she felt her skills offered real value in helping the charity's mission and it's service users.


What is a clinical trial?
Learn more about the science behind clinical trials. How are they planned and run, and by who? Why are they so important?
Funding disclosure
The following companies have provided funding for our Clinical Trials Support Service, but have had no input into service content and delivery:
Autolus, BeiGene, Bristol Myers Squibb, Eli Lilly, Kyowa Kirin, Novartis, Pfizer and Takeda.
Read about how we work with the pharmaceutical industry.
